Knowledge Base • 15 Min Read
Cold Forming Foil: Why Your Pharma Client Rejected the Cheaper PVC Blister
Table of Contents
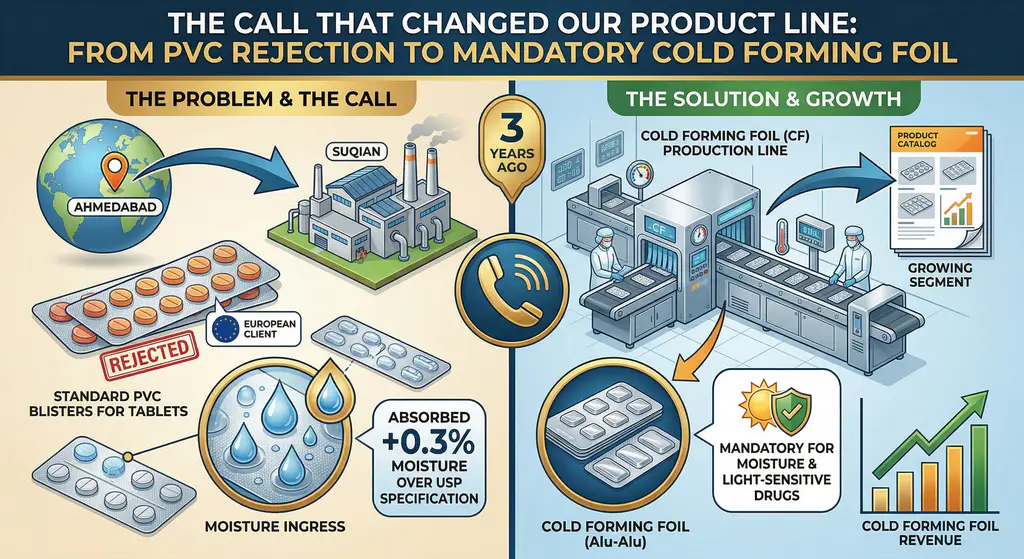
The Call That Changed Our Product Line
Three years ago, a pharmaceutical packaging converter in Ahmedabad called our Suqian factory with a problem that caught us off guard. They had been buying standard PVC thermoform films for years. Suddenly, their biggest client — a European generics manufacturer — rejected an entire shipment of blister-packed tablets. The reason? Moisture ingress. The tablets had absorbed 0.3% more moisture than the USP specification allowed, and the PVC blisters were to blame.
That phone call pushed us to develop our cold forming foil line. Today, it’s one of the fastest-growing segments in our product catalog, and for good reason: when a drug is moisture-sensitive or light-sensitive, cold forming foil isn’t optional — it’s mandatory.
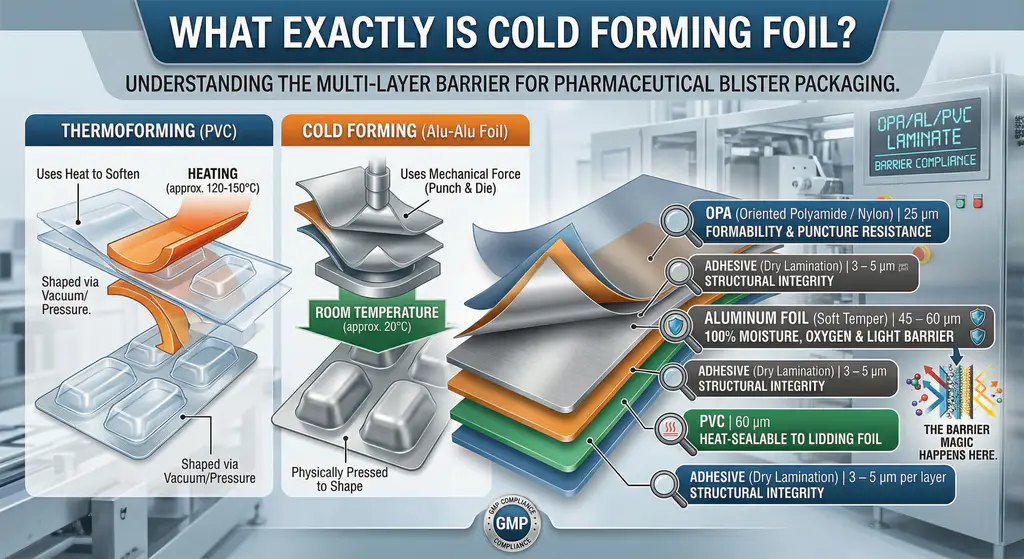
What Exactly Is Cold Forming Foil?
The Layer Structure (This Is Where the Magic Happens):
| Layer | Material | Thickness | Function |
|---|---|---|---|
| Outer | OPA (Oriented Polyamide / Nylon) | 25 μm | Formability & puncture resistance |
| Middle | Aluminum Foil (Soft Temper) | 45 – 60 μm | 100% moisture, oxygen & light barrier |
| Inner | PVC | 60 μm | Heat-sealable to lidding foil |
| Bonding | Adhesive (Dry Lamination) | 3 – 5 μm per layer | Structural integrity |
Cold Forming vs. Thermoforming: The Real Difference
Everyone asks: “What is the difference between cold forming and thermoforming?” Here’s the blunt answer from someone who manufactures materials for both.
Thermoforming uses transparent PVC or PVDC film. It’s fast, cheap, and the patient can see the pill through the blister. But PVC is a terrible moisture barrier. Its Water Vapor Transmission Rate (WVTR) sits around 2.0 – 5.0 g/m²/day. For a standard ibuprofen tablet, that’s fine. For a hygroscopic API like Losartan or a biologic capsule, it’s a death sentence for your shelf life.
Cold forming foil delivers a WVTR of essentially zero — because aluminum is an absolute barrier. No moisture, no oxygen, no UV light gets through. Period.
The Trade-Off Nobody Wants to Hear:
| Factor | Thermoforming (PVC/PVDC) | Cold Forming (Alu-Alu) |
|---|---|---|
| Barrier Performance | Moderate (WVTR: 2-5 g/m²/day) | Absolute (WVTR: ~0) |
| Material Cost | Low ($3-5 /kg) | High ($8-12 /kg) |
| Production Speed | Compact (tight cavity forming) | 15-25 cycles/min |
| Pack Size | Adhesive (Dry Lamination) | 20-30% larger (corner radius limits) |
| Product Visibility | Yes (transparent) | No (opaque) |
Cold forming is slower and more expensive. There’s no way around it. But when your drug costs $50 per tablet and a stability failure means a $2 million recall, that extra $0.03 per blister suddenly looks like the best insurance money can buy.
What Is Cold Form Blister Foil in Pharma? (The Regulatory Reality)
- Hygroscopic APIs: Drugs that absorb moisture from the air (e.g., Aspirin, Ranitidine, certain ARBs).
- Effervescent Tablets: Even trace moisture triggers premature activation.
- Biologics & Peptides: Protein-based drugs that denature with moisture exposure.
- Light-Sensitive Compounds: The opaque aluminum blocks 100% of UV and visible light.

The Forming Process: Where Operators Fail
The “cold” in cold forming foil doesn’t mean “easy.” The mechanical forming process puts enormous stress on the aluminum layer. If your tooling isn’t precise, you’ll get:
- Corner Cracking: The #1 defect. When the punch radius is too tight, the aluminum fractures at the cavity corners, creating micro-channels for moisture ingress. We recommend a minimum corner radius of 3mm for 45μm foil.
- Wrinkling: Caused by excessive material draw. The OPA (nylon) layer is designed to manage this, but cheap nylon with inconsistent molecular orientation will wrinkle under stress.
- Delamination: If the dry-bond adhesive between layers isn’t cured properly, the aluminum separates from the PVC under forming pressure.
Our hot stamping foil articles discuss adhesion physics in depth — the same principles of release force, surface energy, and adhesive chemistry apply here, just in a completely different application context.
How We Test Before We Ship
In our Suqian facility, cold forming foil goes through five quality gates before it leaves the loading dock:
- Pinhole Test: Light table inspection at 100% coverage. One pinhole = rejected roll.
- Formability Test: We stamp test cavities at the customer’s machine specifications and inspect for corner cracks under 50x magnification.
- Peel Strength Test: The bond between OPA/Al and Al/PVC must exceed 3.5 N/15mm. Below that, delamination risk during forming increases exponentially.
- WVTR Verification: We run a 24-hour moisture transmission test on formed samples — not flat samples, because forming stretches the material and can introduce micro-defects.
- Heat Seal Integrity: The PVC inner layer must seal cleanly to standard pharmaceutical lidding foil at 180-220°C without burning the OPA outer layer.
Why Liangyu Foil for Your Pharma Packaging?
- Custom Thickness: 45μm for standard blisters, 60μm for deep-draw cavities (syringes, device trays).
- Certified Production: Our facility operates under ISO quality management with full batch traceability.
- Global Supply Chain: We serve pharmaceutical converters across India, Turkey, Egypt, and Southeast Asia with stable lead times.
The Decision Framework: A Checklist from Our Engineering Desk
Stop overthinking it. Answer these four questions:
- Is your press inline (flexo/offset)? → Cold foil.
- Do you need embossed texture or mirror finish? → Hot foil.
- Is your run under 5,000 units? → Cold foil (lower tooling cost).
- Are you stamping on leather, fabric, or textured board? → Hot foil. Always.
FAQ: From the Pharma Packaging Desk
Can cold forming foil be used for non-pharmaceutical products?
Yes. We’ve supplied cold forming foil for high-end electronics components and sensitive diagnostic test strips — anywhere absolute moisture protection is needed.
What is the shelf life of drugs in cold form blisters vs. PVC?
Depending on the API, cold form packaging can extend shelf life by 12-24 months compared to standard PVC thermoforming. Always validate with your own stability studies.
Is cold forming foil recyclable?
The multi-layer laminate structure (OPA/Al/PVC) makes recycling challenging. However, the industry is actively developing mono-material alternatives. Our Matte Pigment Foil page discusses sustainability trends in foil manufacturing.
The $0.03 Decision That Protects a $50 Drug
Cold forming foil is not the cheapest blister material. It never will be. But for moisture-sensitive, light-sensitive, or high-value pharmaceuticals, it’s the only material that delivers an absolute barrier — and the peace of mind that comes with it.





